Congratulations to the REACH investigators listed below for their recent published work!
- BACPAC REACH
- Nov 8, 2022
- 3 min read
Updated: Nov 9, 2022
Dr. Irina Strigo et al. published a paper "Understanding Pain and Trauma Symptoms in Veterans From Resting-State Connectivity: Unsupervised Modeling. Front Pain Res (Lausanne)." Chronic pain is a major public health concern, especially among Veterans. Chronic pain almost never shows as a single disorder but often overlaps with other conditions, such as trauma, depression, etc. We wanted to examine brain connectivity at rest in a diagnostically heterogeneous samples of Veterans with a range of back pain and trauma symptoms. We used statistical approach that was sensitive to clinical heterogeneity in our sample.
Read the full article in the Frontiers in Pain Research here.

Figure: Multiple biotypes among Veterans with pain and trauma
A) Unsupervised GIMME identified common and unique effective connectivity for the three biotypes based on effective connectivity paths at rest. Biotype 1 [BT1, yellow, n=16] was characterized by functional connectivity to the insula from NAc, ACC and PCC, as well as from thalamus to ACC and bidirectional interconnections between thalamus and PCC. Biotype 2 [BT2, indigo, n=23] was characterized by functional connectivity from the ACC and NAc to thalamus and from ACC to PCC; Biotype 3 [BT3, skyblue, n=15] was characterized by functional connectivity from thalamus and PCC to NAc. Both BT2 and BT3 were characterized by overlapping functional connectivity from insula to ACC, while BT1 and BT3 had overlapping functional connections from PCC to ACC. No connections were shared between USG1 and USG2. (bottom) Graphic representation of clinical and psychological variables among the three neural biotypes identified by unsupervised GIMME based on effective connectivity paths at rest.
B) NAc-Insula connections that were unique to the low pain and trauma symptoms biotype (yellow) was inversely related to pain impact scores (from Brief Pain Inventory) suggesting that such connection may be protective (Pearson’s correlations scatter plot with 95% confidence interval is shown for demonstration). INS=insula cortex, NAc=nucleus accumbens. Severity=pain severity, Interference = pain interference, PCL – PTSD checklist; CES – combat exposure scale; BDI2 – Beck Depression Inventory; PCS – pain catastrophizing scale; PASS20 – pain anxiety symptom scale.
Our approach automatically subgrouped Veterans into three unique biotypes based on their brain connections, regardless of their self-reported pain and trauma levels. Despite demographic and diagnostic similarities, we found neurobiologically dissociable biotypes with different mechanisms for managing pain and trauma.
We believe it is important to study “real” patient and understand how symptom heterogeneity maps onto underlying neural mechanisms. To develop a comprehensive neurobiological model that is needed for effective treatment development, it is necessary to identify the objective mechanisms behind the clinical presentation of pain and trauma – both of these are measured using subjective report. Understanding that the same symptom can be mapped to a different functional connection in the brain has implications for the determination of appropriate biotype-specific interventions that target these neurological systems.
Limitations: This is the first study thus replicating our results is important. The cross-sectional design restricted our ability to draw conclusions as to whether the brain findings represent a vulnerability to, or impact of, chronic pain and/or trauma. This may impact preventative treatment development. Funded in part by the Department of Veterans Affairs.
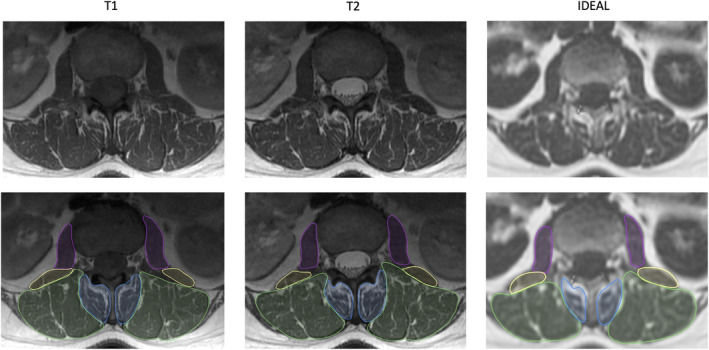
Another publication by Postdoctoral Scholar Dr. Noah Bonnheim and PI Dr. Aaron Fields titled "The contributions of cartilage endplate composition and vertebral bone marrow fat to intervertebral disc degeneration in patients with chronic low back pain."
Read the full article in the European Spine Journal here.
Intervertebral disc degeneration likely causes chronic low back pain in a subset of people, and this understanding has motivated the development of experimental intradiscal biologic therapies designed to regenerate the disc. Factors influencing disc degeneration and the disc’s regenerative potential are not fully understood, but could involve subchondral bone marrow vascularity and/or cartilage endplate (CEP) permeability, since these factors influence the disc’s nutrient supply. In our study, we used imaging biomarkers from quantitative MRI to establish how CEP biochemical composition (surrogate for permeability; measured using T2* relaxation times from ultrashort echo time MRI) and vertebral bone marrow fat fraction (surrogate for perfusion; measured using water-fat MRI) relate to disc degeneration in patients with chronic low back pain. Our results provide the first in vivo evidence that disc degeneration severity relates to compositional deficits in the CEP: we found that CEPs with lower T2* values—implying a fibrotic composition and lower permeability—were associated with more severe disc degeneration both before and after accounting for patient age, sex, and BMI. Conversely, higher bone marrow fat fraction values in the vertebra—reflecting less hematopoietic marrow and decreased perfusion—were not associated with disc degeneration after accounting for patient characteristics. These findings suggest that poor CEP composition plays a significant role in disc degeneration severity in patients with chronic low back pain. Non-invasive assessment of CEP composition with ultrashort echo time MRI represents a promising approach for selecting which degenerated discs are most likely to benefit from intradiscal biologic therapies.



Comments